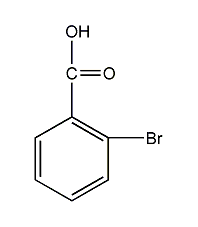
Structural formula
| Business number | 01ZU |
|---|---|
| Molecular formula | C7H5BrO2 |
| Molecular weight | 201.02 |
| label |
2-bromobenzoic acid, o-bromobenzoic acid, o-bromobenzoic acid, 2-Bromobenzoic acid, BrC6H4CO2H |
Numbering system
CAS number:88-65-3
MDL number:MFCD00002402
EINECS number:201-848-6
RTECS number:DG4448035
BRN number:971266
PubChem number:24850027
Physical property data
1. Properties: Colorless monoclinic prismatic crystal.
2. Density (g/mL, 25/4℃): 1.929
3. Relative vapor density (g/mL, air=1): Undetermined
3. p>
4. Melting point (ºC): 150
5. Boiling point (ºC, normal pressure): sub
6. Boiling point (ºC, 5.2kPa): Undetermined
7. Refractive index: Undetermined
8. Flash point (ºC): Undetermined
9. Specific rotation (º): Undetermined
10. Autoignition point or ignition temperature (ºC): Undetermined
11. Vapor pressure (kPa, 25ºC): Undetermined
12. Saturated vapor Pressure (kPa, 60ºC): Undetermined
13. Heat of combustion (KJ/mol): Undetermined
14. Critical temperature (ºC): Undetermined
15. Critical pressure (KPa): Undetermined
16. Log value of oil-water (octanol/water) partition coefficient: Undetermined
17. Explosion upper limit (%, V/V): Undetermined
18. Lower explosion limit (%, V/V): Undetermined
19. Solubility: Soluble in ethanol, ether, acetone, chloroform and Hot water, slightly soluble in cold water.
Toxicological data
None
Ecological data
None
Molecular structure data
1. Molar refractive index: 40.87
2. Molar volume (cm3/mol): 118.1
3. Isotonic specific volume (90.2K ): 319.8
4. Surface tension (dyne/cm): 53.7
5. Polarizability (10-24cm3): 16.20
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 2
4. Number of rotatable chemical bonds: 1
5. Number of tautomers: none
6. Topological molecule polar surface area 37.3
7. Number of heavy atoms: 10
8. Surface charge: 0
9. Complexity: 136
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters�:0
12. Uncertain number of stereocenters of atoms: 0
13. Determined number of stereocenters of chemical bonds: 0
14. Uncertain chemical bonds Number of stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
None
Storage method
This product should be kept sealed.
Synthesis method
Add 0.8L water and 210g (1.5mol) powdered anthranilic acid into the beaker and stir into a paste. While stirring, slowly add cold dilute sulfuric acid composed of 90ml (160g, 1.6mol) concentrated sulfuric acid and 100ml water (this can avoid agglomeration), cool externally, and stir thoroughly for 10 minutes. Slowly add a cold solution of 105g (1.5mol) NaNO2 dissolved in 150mL water at 5~12°C. The volume after addition is about 1.4L and is basically clear; add 5g urea and stir for 10 minutes to decompose excess nitrous acid (add if necessary 1 mL of octanol to eliminate foam) and leave for 10 minutes to allow a little undiazotized anthranilic acid to settle (longer standing and cooling will precipitate crystallized sodium sulfate). Add this diazonium salt solution in portions to the copper bromide solution at about 80°C. Add 240g (1.65mol) cuprous bromide dissolved in 600mL 42% hydrobromic acid (4.4mol) into a 5L beaker, and heat the reaction to 80°C while stirring. When cooled to 60°C, filter out (retain the mother liquor), use 150 mL of hot water to elute the cuprous mother liquor and combine the remaining solutions; wash thoroughly with hot water until the blue color is almost free of copper salts. Stir the wet product with 1.2L hot water into a paste, neutralize and dissolve with 40% NaOH (the reaction product turns from gray-green to yellow-brown as the end point, pH9~10), heat to 80°C, filter, the filtrate will be dark gray; then Heat to 80℃, slowly add 20g of hot concentrated solution of KMnO4 while stirring, filter out MnO2 and rinse twice with warm water. Combine the brown filtrate and washing liquid, heat to 90℃ and acidify with hydrochloric acid while stirring, maintain 90℃ , stir for 10 minutes, cool to 60°C, filter out, and wash thoroughly with hot water at about 60°C until the washing liquid is only very light yellow, and obtain 400g of dark yellow wet product. After drying, 245g of product was obtained, with a melting point of 146.5~147.6℃.
Refined: Dissolve the above 245g crude product in 270mL hot acetic acid (1:1.1), decolorize and filter, cool to 20℃ (stir several times after crystallization begins to avoid agglomeration), filter out, and rinse once with a little acetic acid. 220g (wet) product with a brownish-yellow appearance was obtained; then it was dissolved, precipitated, washed once as above, and air-dried to obtain 175g of product. 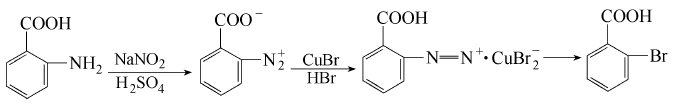
Purpose
Used in organic synthesis.

 微信扫一扫打赏
微信扫一扫打赏

