Background and overview[1]
The chemical formula of crystalline sodium sulfide is Na2S·9H2O, and the formula weight is 240.18. Colorless crystal, easily deliquescent, soluble in water and ethanol. After hydrolysis, the solution is alkaline and corrosive. When it comes into contact with skin or cutin, it can soften and dissolve it and cause hair to fall off. Dissolved in crystal water at 50°C. It turns yellow when exposed to light and can be oxidized into sodium thiosulfate in the air. When sulfur is added to an aqueous solution, sodium polysulfide (Na2Sx, x=2, 3, 4 or 5) is generated. Used in the preparation of sulfur dyes, leather hair removers, and as reducing agents in the organic chemical industry. By mixing and calcining sodium sulfate with carbon, sodium sulfate can be reduced to form sodium sulfide. Through experiments, it was found that the oxidation product of crystalline sodium sulfide should be a mixture of elemental sulfur, sodium polysulfide, sodium sulfite, and sodium sulfate. In experiments, the content of partial oxidation products is relatively small, so it is easy to be ignored.
Preparation[2]
1) Process carbon dioxide to produce barium carbonate: Use industrial barium sulfate calcined material as raw material, dissolve it in water and stir it to separate the solid and liquid to make a cleaner barium sulfide solution of about 20%. The mixed gas of carbon dioxide and hydrogen sulfide is countercurrently contacted with the barium sulfide solution in the carbon dioxide absorption tower to generate product barium carbonate and hydrogen sulfide gas. The chemical equation for barium sulfide to remove carbon dioxide is as follows:
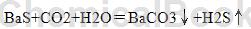
The solid-liquid mixture after generating barium carbonate flows out from the bottom of the carbon dioxide absorption tower and enters the barium carbonate solution storage tank. After separation by a centrifuge, the mother liquor is further dissolved in the liquid preparation process to form a barium sulfide solution, which is recycled back and forth. . The separated barium carbonate solid is dried and then fed into the silo for packaging of finished products.
2) Use hydrogen sulfide treated with barium sulfide to produce sodium sulfide
The treated relatively clean hydrogen sulfide gas reacts with the caustic soda solution to generate sodium sulfide. After evaporation and crystallization separation, sodium sulfide crystals with higher purity can be obtained. The reaction equation is as follows:
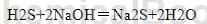
Apply[3]
Examples of applications of crystalline sodium sulfide are as follows: Preparing a film-grade polyphenylene sulfide resin, which involves the technical field of polymer material synthesis. The preparation method includes: adding N-methylpyrrolidone, crystalline sodium sulfide, and anhydrous lithium chloride to the reaction kettle, and performing temperature-raising dehydration under nitrogen protection; adding crystalline sodium sulfide to the reaction kettle; adding N to the reaction kettle – After the addition of methylpyrrolidone, p-dichlorobenzene, and m-dichlorobenzene, replace the air in the reaction kettle with nitrogen, and then seal the reaction kettle; heat the reaction kettle to 230°C to perform preliminary polymerization, and then heat the reaction kettle to 279°C for late-stage polymerization; after the reaction is completed and cooled, the slurry in the reaction kettle is filtered, washed, and dried to obtain a polyphenylene sulfide resin product. By adding a certain amount of sodium sulfide to the reactor and performing a polymerization reaction, the production capacity of the reactor per unit volume is increased, solvent consumption and energy consumption are reduced, production costs are reduced, and the polyphenylene sulfide resin produced can reach the film level Resin requirements.
Main reference materials
[1] Practical Chemistry Dictionary for Middle School Teachers
[2] CN201010596847.2 A waste gas treatment process and device containing hydrogen sulfide and carbon dioxide
[3] CN201710705207.2 Preparation method of film-grade polyphenylene sulfide resin

 微信扫一扫打赏
微信扫一扫打赏

