Background and overview[1][2]
3-(2-Chloro-6-fluorophenyl)-5-methylisoxazole-4-carboxylic acid is a key intermediate for the antibiotic flucloxacillin sodium. Also known as Flucloxacillin – Impurity D.
The existing chemical synthesis method of 3-(2-chloro-6-fluorophenyl)-5-methylisoxazole-4-carboxylic acid uses phosphorus oxychloride as the chlorination reagent, which is harmful to the equipment. The corrosion is serious, and the large amount of phosphorus-containing wastewater in the products obtained by the process is difficult to treat. The pollution problem is serious and the environmental problem is outstanding. The exhaust gas from the thionyl chloride process contains a large amount of asphyxiating sulfur dioxide (one of the six indicators of strict control of the atmosphere by the national environmental protection). The treatment of the three wastes is difficult and expensive. In addition, the transportation and use of thionyl chloride are strictly controlled, which requires high sealing performance of reaction equipment and requires a large investment. The production safety is poor and the reaction cycle is long.
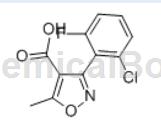
3-(2-Chloro-6-fluorophenyl)-5-methylisoxazole-4-carboxylic acid
Apply[3]
Can prepare 3-(2-chloro-6-fluorophenyl)-5-methylisoxazole-4-isoxazolecarboxyl chloride:
3-(2-Chloro-6-fluorophenyl)-5-methylisoxazole-4-carboxylic acid:bis(trichloromethyl)carbonate:tetramethylurea=1:0.33:0.02 (Molar ratio), the input amount of 3-(2′-chloro-6′-fluorophenyl)-5-methyl-4-isoxazolecarboxylic acid is 25.55g (100mmol), bis(trichloromethyl)carbonate The input amount is 9.80g (33mmol), the catalyst amount is 0.232g (2mmol), and the toluene amount is 3-(2′-chloro-6′-fluorophenyl)-5-methyl-4-isoxazolecarboxylic acid mass. 10 times.
Add 3-(2′-chloro-6′-fluorophenyl)-5-methyl-4-isoxazolecarboxylic acid, tetramethylurea and toluene into the reaction kettle, stir evenly, and stir at room temperature. Add the toluene solution of bis(trichloromethyl)carbonate dropwise within 45 minutes, and turn on the hydrogen chloride absorption system at the same time. Then raise the temperature to 110°C and reflux for 2 hours. After the reaction is completed, the toluene is recovered by distillation under reduced pressure, and finally collected at 0.667KPa. The fraction at 168~170℃ solidifies under freezing.
26.22 grams of product was obtained, with a yield of 95.7%, a melting point of 48-52°C, and a content (GC) of 99.9%. 1H-NMR (CDCl3) δ: 2.87 (3H, s), 7.08 (1H, t, J=8.8Hz), 7.33 (1H, t, J=8.0Hz), 7.41-7.47 (1H, m).
Main reference materials
[1] Chen Zhiwei, Yan Weihua, & Su Weike. (2008). Synthesis of 3-(2′-chloro-6′-fluorophenyl)-5-methyl-4-isoxazolecarbonyl chloride. Chinese Modern Applied Pharmacy, 25(4), 308-309.
[2] Dai Liyan, Chen Yingqi, & Wu Zhaoli. (2001). Temafloxacin intermediate 7-chloro-6-fluoro-1-(2,4-difluorophenyl)-1,4-di Improvement of the synthesis method of hydrogen-4-oxy-3-quinolinecarboxylic acid. Journal of Chemical Engineering in Colleges and Universities, 15(5), 468-471.
[3] Zhou Gaiping. (2013). Synthesis process of flucloxacillin sodium. Shanxi Medical Journal (12), 1429-1430.

 微信扫一扫打赏
微信扫一扫打赏

