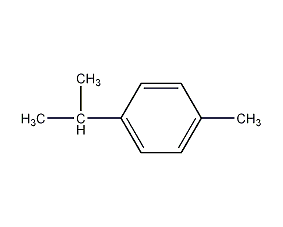
Structural formula
| Business number | 02GM |
|---|---|
| Molecular formula | C10H14 |
| Molecular weight | 134.22 |
| label |
1-Methyl-4-isopropylbenzene, 1-isopropyl-4-methylbenzene, paracymene, 1-Isopropyl-4-methylbenzene, thinner, hydrocarbon solvents, Edible spices |
Numbering system
CAS number:99-87-6
MDL number:MFCD00008893
EINECS number:202-796-7
RTECS number:GZ5950000
BRN number:1903377
PubChem number:24858092
Physical property data
1. Properties: Colorless, transparent and flammable liquid with aromatic odor and irritation.
2. Boiling point (ºC, 101.3kPa): 177.10
3. Melting point (ºC): -67.935
4. Relative density (g/mL, 20/4ºC): 0.8573
5. Relative density (g/mL, 25/4ºC): 0.8533
6. Relative vapor density (g/mL, air=1): 4.7
7. Refractive index (n20D): 1.4909
8. Refractive index (25ºC): 1.4885
9. Viscosity (mPa·s, 20ºC): 3.402
10. Viscosity (mPa·s, 30ºC): 1.600
11. Flash point (ºC, closed): 47.2
12. Fire point (ºC): 436.1
13. Heat of fusion (KJ/mol) : -9.667
14. Heat of evaporation (KJ/mol, 25ºC): -50.33
15. Heat of evaporation (KJ/mol, b.p.): -38.19
16. Heat of formation (KJ/mol): -9.667
17. Specific heat capacity (KJ/(kg·K),-23.9ºC, constant pressure): 1.76
18 . Critical temperature (ºC): 380
19. Critical pressure (MPa): 2.84
20. Boiling point rising constant (ºC/mol·1000g): 5.52
21. Conductivity (S/m, 25ºC): 2×10-8
22. Solubility (%, water): 0.034
23. Vapor pressure (kPa, 25ºC): 0.20
24. Vapor pressure (kPa, 79.2ºC): 4.00
25. Vapor pressure (kPa, 109.1ºC): 13.33
26. Lower explosion limit (%, V/V): 0.7
27. Upper explosion limit (%, V/V): 5.6
28. Solubility : Insoluble in water, soluble in ethanol, ether, acetone, chloroform and other organic solvents.
Toxicological data
1. Skin/eye irritation: Standard Dresser test: rabbit skinIn contact, 500mg/24HREACTION SEVERITY, medium reaction; 2. Acute toxicity: rat oral LD50: 4750mg/kg; 3. Toxicity is similar to toluene, with irritating effect, and inhalation of its liquid can cause chemical pneumonitis. It is a primary skin irritant and long-term exposure at low concentrations can cause skin dryness, degreasing and erythema.
Ecological data
This substance may be harmful to the environment. It is recommended not to let it enter the environment. It may cause pollution to water bodies and the atmosphere.
Molecular structure data
1. Molar refractive index: 45.26
2. Molar volume (cm3/mol): 155.7
3. Isotonic specific volume (90.2K ): 359.9
4. Surface tension (dyne/cm): 28.5
5. Dielectric constant: 2.34
6. Dipole moment (10-24cm3):
7. Polarizability: 17.94
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 0
4. Number of rotatable chemical bonds: 1
5. Number of tautomers: none
6. Topological molecule polar surface area 0
7. Number of heavy atoms: 10
8. Surface charge: 0
9. Complexity: 86.2
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. Avoid contact with strong oxidants. 2. Oxidation produces p-toluic acid, p-methylacetophenone, p-isopropylbenzoic acid and terephthalic acid. Oxidation with alkaline potassium permanganate produces p-hydroxybenzoic acid. Hydrogenation produces para-alkanes. Heating and cracking produces benzene, toluene, naphthalene, anthracene, etc. Treatment with aluminum trichloride produces diisopropylbenzene, toluene, xylene and 1-methyl-3,5-diisopropylbenzene. Carvacrol (2-methyl-5-isopropylphenol), thymol (5-methyl-2-isopropylphenol), and menthol are synthesized through halogenation, nitration, and sulfonation reactions. 3. Insoluble in water, soluble in ethanol, ether, acetone, chloroform and other organic solvents. The boiling point is similar to that of turpentine, but its solubility is greater than that of turpentine. Can dissolve grease, resin, rubber, etc. It will change color after being left for a long time and is non-corrosive to metals. However, it has a rapid effect on rubber, so avoid using it in valves and gaskets. In case of fire, use foam fire extinguishing agent, carbon dioxide, dry chemical fire extinguishing agent, carbon tetrachloride, etc. to extinguish the fire. 4. Found in flue-cured tobacco leaves.
Storage method
Store in a cool, ventilated warehouse. Keep away from fire and heat sources. The storage temperature should not exceed 30℃. should be kept away from oxidizer, do not store together. Use explosion-proof lighting and ventilation facilities. It is prohibited to use mechanical equipment and tools that are prone to sparks. The storage area should be equipped with emergency release equipment and suitable containment materials. It should be stored in a cool place away from light and sealed. Can be stored in iron, mild steel, copper or aluminum containers.
Synthesis method
1. Obtained from the reaction of camphor and phosphorus pentoxide through nitrification. First, heat powdered camphor and phosphorus pentoxide at 176°C for 12 hours to dehydrogenate to generate isopropyl-p-toluene. After acid and alkali refining and vacuum distillation, the finished product was obtained with a yield of 37%.
2. It is formed by dehydrogenating d-limonene or dipentene.
Purpose
Used for organic synthesis and preparation of paint thinners. As raw material for organic synthesis. Mixtures with ethanol, butanol, and acetone are solvents and thinners for paints, varnishes, nitrocellulose sprays, greases, and resins.

 微信扫一扫打赏
微信扫一扫打赏

