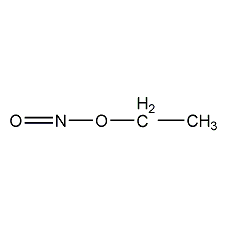
Structural formula
| Business number | 0307 |
|---|---|
| Molecular formula | C2H5NO2 |
| Molecular weight | 75.07 |
| label |
Ethyl nitrite alcohol, Nitrousethylether, food additives, flavor enhancer, pesticides |
Numbering system
CAS number:109-95-5
MDL number:MFCD00010257
EINECS number:203-722-6
RTECS number:None
BRN number:None
PubChem number:24858758
Physical property data
1. Properties: gas or colorless to light yellow clear liquid with special odor, sweet taste and extremely volatile. [1]
2. Melting point (℃): -50[2]
3. Boiling point (℃): 17.2[3]
4. Relative density (water=1): 0.90 (15.5℃)[4]
5. Relative vapor density (air = 1): 2.59[5]
6. Heat of combustion (kJ/mol): -1351.3[6]
7. Octanol/water partition coefficient: 1.37[7]
8. Flash point (℃): -35 (CC)[8]
9. Ignition temperature (℃): 90 (decomposition) [9]
10. Explosion limit ( %): 50[10]
11. Lower explosion limit (%): 3.0[11]
12. Dissolution Properties: Insoluble in water, miscible in ethanol and ether. [12]
Toxicological data
1. Acute toxicity[13] LC50: 160ppm (rat inhalation, 4h)
2. Irritation No information yet
Ecological data
1. Ecotoxicity No data available
2. Biodegradability No data available
3 .Non-biodegradability No information available
4. Other harmful effects[14] This substance is harmful to the environment and should be specially Pay attention to water pollution.
Molecular structure data
1. Molar refractive index: 16.86
2. Molar volume (cm3/mol): 71.5
3. Isotonic specific volume (90.2K ): 168.6
4. Surface tension (dyne/cm): 30.8
5. Polarizability (10-24cm3): 6.68
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): 0.4
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 3
4. Number of rotatable chemical bonds: 1
5. Number of tautomers: none
6. Topological molecule polar surface area 38.7
7. Number of heavy atoms: 5
8. Surface charge: 0
9. Complexity: 28.8
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. StabilityProperties[15] Stable
2. Incompatible materials [16] Strong oxidizing agent p>
3. Conditions to avoid contact[17] Heat and light
4. Polymerization hazard[18 ] No polymerization
5. Decomposition products[19] Nitrogen oxides
Storage method
Storage Precautions[20] Store in a cool, well-ventilated special warehouse, and implement the “two people to receive and receive, two people to keep” system. Keep away from fire and heat sources. Keep away from light. The storage temperature should not exceed 29°C. Keep container tightly sealed. should be kept away from oxidizer, do not store together. Use explosion-proof lighting and ventilation facilities. It is prohibited to use mechanical equipment and tools that are prone to sparks. The storage area should be equipped with emergency release equipment and suitable containment materials.
Synthesis method
1. Obtained from the reaction of ethanol and sodium nitrite. Dissolve sodium nitrite in hot water, add ethanol after cooling, slowly inject sulfuric acid, that is, ethyl nitrite gas will be generated, and pass it into ethanol cooled with ice brine for absorption to obtain an ethanol solution of ethyl nitrite.
2. Its preparation method is to obtain ethyl nitrite by reacting ethanol and sodium nitrite. C2H5OH+NaNO2[H2SO4]→C2H5ONO Dissolve sodium nitrite in hot water, add ethanol after cooling, slowly inject sulfuric acid, that is, ethyl nitrite gas will be generated, pass it into ethanol cooled with ice salt water for absorption, and obtain nitrous acid For the ethanol solution of ethyl ester, the ethyl nitrite content in ethanol is about 30% according to the weight gain method.
3. Sodium nitrite is replaced with sulfuric acid in an aqueous solution to form acid, and then esterified with ethanol present in the solution.
Purpose
1. α-oxime sodium benzene acetonitrile, an intermediate used in the synthesis of organophosphorus pesticide phoxim in pesticide production, and used in the pharmaceutical industry to prepare pralidoxime chloride.
2. Used in the pharmaceutical industry as an intermediate in organic synthesis. [21]

 微信扫一扫打赏
微信扫一扫打赏

