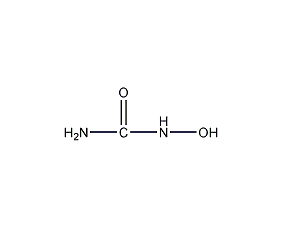
Structural formula
| Business number | 03KL |
|---|---|
| Molecular formula | CH4N2O2 |
| Molecular weight | 76.06 |
| label |
Carbamylhydroxylamine, hydroxyurea, N-Hydroxyurea, carbamoylhydroxylamine, aliphatic compounds |
Numbering system
CAS number:127-07-1
MDL number:MFCD00007943
EINECS number:204-821-7
RTECS number:YT4900000
BRN number:1741548
PubChem number:24278474
Physical property data
1. Character:White needle crystal
2. Melting point (℃):142-146
3. Water solubility:soluble
Toxicological data
None
Ecological data
None
Molecular structure data
5. Molecular property data:
1. Molar refractive index: 15.37
2. Molar volume (m3/mol):52.1
3. isotonic specific volume (90.2K):150.7
4. Surface Tension (dyne/cm):69.7
5. Polarizability(10-24cm3):6.09
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 3
3. Number of hydrogen bond acceptors: 2
4. Number of rotatable chemical bonds: 0
5. Number of tautomers: 4
6. Topological molecule polar surface area 75.4
7. Number of heavy atoms: 5
8. Surface charge: 0
9. Complexity: 42.9
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
basic properties
White needle-like crystals. Melting point: 70-72℃ (decomposition) or 141℃ decomposition. Easily soluble in water and hot ethanol, slightly soluble in cold ethanol, insoluble in ether; benzene. Not stable in contact with water or heat. Odorless and tasteless
Storage method
storage
Stored sealed in a cool, dry place.
Synthesis method
Brief description of production methods
Obtained from the reaction of ethyl carbamate and hydroxylamine hydrochloride. Cool the sodium hydroxide solution to 20-25°C. Add ethyl carbamate and hydroxylamine hydrochloride alternately while stirring, and react at 25-28°C for 16 hours. Neutralize with hydrochloric acid to pH 6.5-7, and control the temperature not to exceed 25°C. Then concentrate under reduced pressure, filter while hot, and cool to below 0°C overnight to precipitate crystals. Filter, wash the crystals with ice water, and dry to obtain crude hydroxyurea. The yield is about 65%. After refining, pharmaceutical grade hydroxyurea can be obtained.
Purpose
Purpose
This product is an anti-tumor drug.
Y: Arial; mso-ascii-font-family: ‘Times New Roman’; mso-hansi-font-family: ‘Times New Roman’; mso-bidi-font-family: Arial”>(10-24cm3): 6.09
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 3
3. Number of hydrogen bond acceptors: 2
4. Number of rotatable chemical bonds: 0
5. Number of tautomers: 4
6. Topological molecule polar surface area 75.4
7. Number of heavy atoms: 5
8. Surface charge: 0
9. Complexity: 42.9
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
basic properties
White needle-like crystals. Melting point: 70-72℃ (decomposition) or 141℃ decomposition. Easily soluble in water and hot ethanol, slightly soluble in cold ethanol, insoluble in ether; benzene. Not stable in contact with water or heat. Odorless and tasteless
Storage method
storage
Stored sealed in a cool, dry place.
Synthesis method
Brief description of production methods
Obtained from the reaction of ethyl carbamate and hydroxylamine hydrochloride. Cool the sodium hydroxide solution to 20-25°C. Add ethyl carbamate and hydroxylamine hydrochloride alternately while stirring, and react at 25-28°C for 16 hours. Neutralize with hydrochloric acid to pH 6.5-7, and control the temperature not to exceed 25°C. Then concentrate under reduced pressure, filter while hot, and cool to below 0°C overnight to precipitate crystals. Filter, wash the crystals with ice water, and dry to obtain crude hydroxyurea. The yield is about 65%. After refining, pharmaceutical grade hydroxyurea can be obtained.
Purpose
Purpose
This product is an anti-tumor drug.

 微信扫一扫打赏
微信扫一扫打赏

