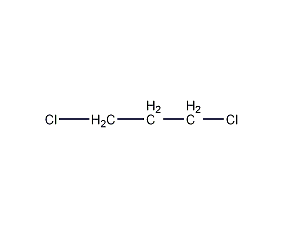
Structural formula
| Business number | 03UX |
|---|---|
| Molecular formula | C3H6Cl2 |
| Molecular weight | 112.99 |
| label |
Dichlorotrimethylene, trimethylene chloride, Trimethylene dichloride, 1,3-Propylenechloride, chromatographic material reference material, detergent, Halogenated hydrocarbon solvents |
Numbering system
CAS number:142-28-9
MDL number:MFCD00000999
EINECS number:205-531-3
RTECS number:TX9660000
BRN number:None
PubChem ID:None
Physical property data
1. Properties: colorless liquid with an odor similar to chloroform. [1]
2. Melting point (℃): -99.5[2]
3. Boiling point (℃): 120~122[3]
4. Relative density (water=1): 1.20[4]
5. Relative vapor density (air=1): 3.9[5]
6. Saturated vapor pressure (kPa): 2.4 (20℃)[6]
7. Heat of combustion (kJ/mol): -1751.8[7]
8. Critical pressure (MPa): 4.23[ 8]
9. Octanol/water partition coefficient: 2.0[9]
10. Flash point (℃): 15.6 ( OC); 21 (CC) [10]
11. Explosion upper limit (%): 14.5[11]
12. Lower explosion limit (%): 3.4[12]
13. Solubility: Slightly soluble in water, easily soluble in most organic solvents such as ethanol and ether. [13]
14. Relative density (25℃, 4℃): 1.1818
15. Refractive index at room temperature (n25): 1.4460
16. Solubility parameter (J·cm-3)0.5: 19.819
17. van der Waals area (cm2·mol-1): 7.650×109
18. van der Waals Volume (cm3·mol-1): 53.930
19. The gas phase standard claims heat (enthalpy) (kJ·mol-1 ): -159.2
20. Liquid phase standard claims heat (enthalpy) (kJ·mol-1): -200.0
21 .Liquid phase standard hot melt (J·mol-1·K-1): 149.6
Toxicological data
1. Acute toxicity[14] LD50: 3600mg/kg (mouse, exposure route unknown)
2. Irritation No data available
3. Mutagenicity[15] Microbial mutagenicity: Salmonella typhimurium 10 μmol/dish. Micronucleus test: human lymphocytes 500 μmol/L. DNA damage: human lymphocytes 500 μmol/L. Sister chromatid exchange: hamster lung 6600 μmol/L.
Ecological data
1. Ecotoxicity[16]
LC50: 84ppm (7d) (guppy); 86.7 mg/L (96h) (red bass); 131mg/L (96h) (fathead minnow); 282mg/L (96h) (water fleas); 10.3mg/L (96h) (sugar shrimp)��
EC50: 72.2mg/L (96h) (green algae)
2. Biodegradability No data yet
3. Non-biodegradability[17]
In the air, when the hydroxyl radical concentration is 5.00×10 5 pieces/cm3, the degradation half-life is 21 days (theoretical).
When the pH value is 7 and the temperature is 25°C, the hydrolysis half-life is 2.3a (theoretical).
4. Other harmful effects[18] This substance is harmful to the environment and can cause pollution to water bodies, especially Bioaccumulation occurs in aquatic organisms.
Molecular structure data
1. Molar refractive index: 25.64
2. Molar volume (cm3/mol): 100.8
3. Isotonic specific volume (90.2K ): 228.3
4. Surface tension (dyne/cm): 26.3
5. Polarizability (10-24cm3): 10.16
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 0
4. Number of rotatable chemical bonds: 2
5. Number of tautomers: none
6. Topological molecule polar surface area 0
7. Number of heavy atoms: 5
8. Surface charge: 0
9. Complexity: 12.4
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. It is flammable when exposed to open flames or high heat. It decomposes when heated to release highly toxic phosgene. It can react with oxidants.
2. Stability[19] Stable
3. Incompatible substances[20] Strong oxidants, acids, alkalis, aluminum
4. Conditions to avoid contact[21] Heating
5. Polymerization hazard[22] No polymerization
6. Decomposition products[23] Hydrogen chloride, phosgene
Storage method
Storage Precautions[24] Stored in a cool, ventilated warehouse. Keep away from fire and heat sources. The storage temperature should not exceed 37°C. Keep container tightly sealed. They should be stored separately from oxidants, acids, alkalis, etc. and avoid mixed storage. Use explosion-proof lighting and ventilation facilities. It is prohibited to use mechanical equipment and tools that are prone to sparks. The storage area should be equipped with emergency release equipment and suitable containment materials.
Synthesis method
Originated from the reaction of propylene glycol and hydrochloric acid.
Purpose
1. Used as organic synthesis intermediates and solvents.
2. Used in organic synthesis, making detergents, and also used as solvents. [25]

 微信扫一扫打赏
微信扫一扫打赏

