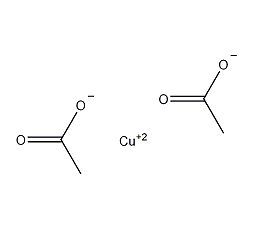
Structural formula
| Business number | 03VA |
|---|---|
| Molecular formula | C4H6CuO4 |
| Molecular weight | 181.63 |
| label |
copper acetate, Anhydrous copper acetate, Cupric acetate, oxidizing agent |
Numbering system
CAS number:142-71-2
MDL number:MFCD00008690
EINECS number:205-553-3
RTECS number:AG3480000
BRN number:None
PubChem number:24882058
Physical property data
1. Character: blue crystal
2. Density (g/mL, 25/4℃): 1.93
3. Melting point (ºC): 130~140
4. Solubility: soluble in water (6.79 g/100 mL, 25 oC), acetic acid and pyridine, insoluble in ether.
Toxicological data
1. Acute toxicity:
Rat oral LD50: 501 mg/kg;
Rat abdominal LD50: 14700 ug/kg;
Rat subcutaneous LD50: 350 mg/kg;
Rat intravenous LD50: 17100 ug/kg;
Mouse oral LD50: 196 mg/kg;
Mouse abdominal LD50: 2500 ug/kg;
Mouse intravenous LD50: 20 mg/kg.
2. Chronic toxicity/carcinogenicity
Rat oral TDLo: 23750 mg/kg/42W-C;
3. Reproductive toxicity:
p>
Rat transdermal TDLo: 40 mg/kg
Ecological data
1. Other harmful effects: This substance may be harmful to the environment, and special attention should be paid to water bodies.
Molecular structure data
None
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 4
4. Number of rotatable chemical bonds: 0
5. Number of tautomers: none
6. Topological molecule polar surface area 80.3
7. Number of heavy atoms: 9
8. Surface charge: 0
9. Complexity: 25.5
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 3
Properties and stability
1. Stable under normal temperature and pressure.
2. Incompatible materials: oxidants, alkali.
3. It is hygroscopic and should be stored in a dry place. Irritating to skin, eyes and respiratory system.
Storage method
Store in a dry and sealed container at room temperature.
Synthesis method
1. Dissolve 50g copper sulfate pentahydrate in 500ml water, filter, and dissolve 57g sodium carbonate decahydrate in 240ml water, heat to 60°C, slowly add copper sulfate solution, and stir continuously. Leave to stand, filter out the precipitate, and wash with hot water until there is no sulfuric acid. Place the precipitate in 300 ml of water, add 2 ml of ammonia water, stir, let stand, pour out the upper solution, and wash and precipitate several times. Add 180ml water to the beaker, heat to 60℃, add 22g glacial acetic acid, and then addWash the above-mentioned basic copper carbonate until there is slightly remaining at the bottom of the container. Filter, and when the filtrate is evaporated and concentrated to 1/3 of the original volume, cool, filter, wash with 2 ml of water, and dry at room temperature to obtain the finished product. The mother liquor continues to evaporate, and 5ml of 25% acetic acid is added when crystallization is about to occur, and some finished products can be obtained. It got 32-35g.
2. Heat copper acetate (II) monohydrate to 105°C, or heat reflux with copper acetate monohydrate and acetic anhydride, cool and filter, and place the resulting crystals in a solid potassium hydroxide or acetic anhydride solution. Dry in concentrated sulfuric acid vacuum desiccator.
3. Anhydrous copper acetate can be prepared by heating at 90 oC or refluxing in acetic anhydride solution and then washing with ether.
Purpose
1. Analytical reagents, organic synthesis catalysts, pesticides, fungicides, printing and dyeing fixatives, and intermediates for preparing Paris Green. Used as chromatographic analysis reagents.
2. Copper acetate can be used as an oxidizing agent for carbanions, free radicals and hydrocarbon compounds, and can realize oxidative coupling reactions of electronegative substrates and Si-C, Bi-C, Pb-C and Solvent cleavage reaction of Sb-C bond. It can also be used in the cyclopropylation reaction of olefins and diazo esters. Copper acetate can also participate in the reaction as a Lewis acid [1].
Copper acetate has oxidative activity towards carbanions, and can realize the oxidative coupling reaction of terminal alkynes in the presence of pyridine to obtain diyne compounds (Formula 1~Formula 4)[2] .
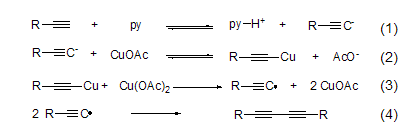
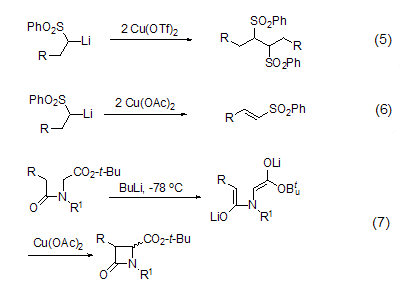
Using trifluoromethanesulfonic acid Using copper as an oxidizing agent can realize the self-coupling reaction of α-sulfonyl lithium carbon anion (Formula 5), but using copper acetate as an oxidizing agent can oxidize it to α,β-unsaturated sulfone compound (formula 6)[3]. Other carbanions can also undergo coupling reactions under the action of copper acetate, such as the synthesis of β-lactam (Formula 7)[4].
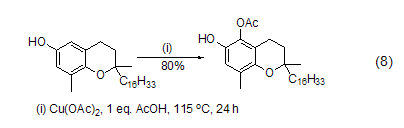
Copper acetate can also oxidize hydrocarbons C-H bonds of the substrate. For example, the ortho-hydroxylation reaction of phenol can be catalyzed by copper acetate in the presence of oxygen and morpholine. When oxygen is isolated, quantitative copper acetate can realize the ortho-acetation reaction of phenol in the presence of acetic acid (Equation 8)[5].
Peroxyester catalyzed by copper acetate It can act on propylene compounds to produce a reaction in which the allyl hydrogen is replaced by an acyloxy group (formula 9)[6]. The reaction is likely to first form an allyl radical, and then rapidly react with Cu(II) to obtain a Cu(III) intermediate, which then undergoes a pericyclic transition state to obtain the final acyloxy-substituted alkene.
Allyl oxidation reactions, such as N-Boc-allylamine intramolecular cyclization reaction (Formula 10), can also be achieved under the combined action of catalytic amounts of palladium acetate and excess copper acetate. [7].
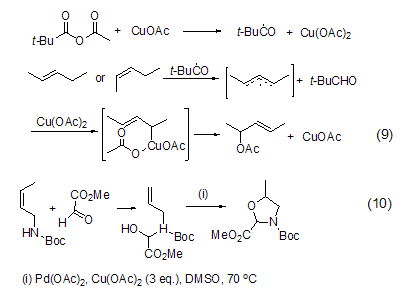
Copper acetate can also achieve carbon- Oxidation reaction of metallic bonds. In an atmosphere of methanol and oxygen, a catalytic amount of copper acetate can promote the cleavage of the vinyl Si-C bond to obtain a vinyl ether compound (formula 11) [8]. The reaction has a high degree of stereoselectivity. Trans-enol ether is completely obtained. The corresponding aldehydes can also be obtained in the presence of water.
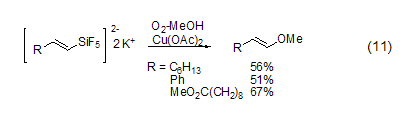
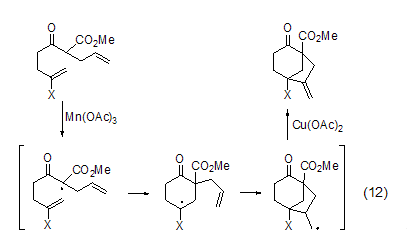
Use in combination with free radical initiator , Copper acetate can also participate in the oxidation reaction of free radicals. For example, β-carbonyl ester obtains free radicals under the oxidation of Mn(OAc)3, and then undergoes intramolecular cyclization to obtain alkyl radicals, which are then oxidized under the action of copper acetate. Oxidation to the corresponding alkene (Formula 12)[9].
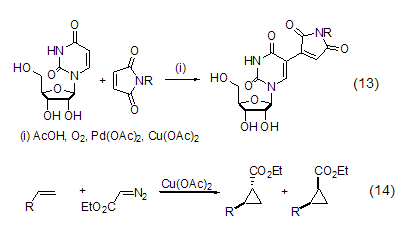
Copper acetate can also be used as a palladium reagent The oxidant in the catalytic reaction re-oxidizes the reduced palladium reagent into Pd(II), and then re-enters the catalytic cycle (Formula 13)[10]. In addition, the combination of copper acetate and chiral imine ligands can induce the asymmetric cyclopropylation reaction of alkenes and diazoacetates (Equation 14)[11].
Copper acetate can react with nitrogen-containing compounds Effectively coordinated, it can participate in the reaction of nitrogen-containing compounds as a Lewis acid. For example, a catalytic amount of copper acetate can convert hydrazide to carboxylic acid in the presence of oxygen (Equation 15)[12].
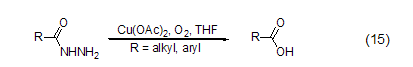
Copper acetate also reacts in the presence of oxygen It can catalyze the coupling reaction of phenylacetylene and dimethylamine to obtain N,N-dimethyl-2-phenylacetylene amine compound (formula 16)[13]. Oxygen is a key factor in this reaction, and in the absence of oxygen, 1,4-diphenylbutyne is the only coupling product.
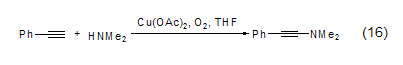

 微信扫一扫打赏
微信扫一扫打赏

