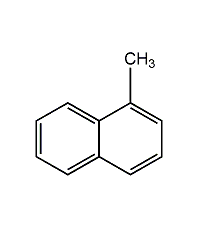
Structural formula
| Business number | 021X |
|---|---|
| Molecular formula | C11H10 |
| Molecular weight | 142.20 |
| label |
α-methylnaphthalene, α-Methylnaphthalene, heat carrier, Surfactant, sulfur extractant, Hydrocarbon solvents |
Numbering system
CAS number:90-12-0
MDL number:MFCD00004034
EINECS number:201-966-8
RTECS number:QJ9630000
BRN number:506793
PubChem number:24901543
Physical property data
1. Properties: colorless oily liquid with an odor similar to naphthalene. [1]
2. Melting point (℃): -22[2]
3. Boiling point (℃): 240~243[3]
4. Relative density (water=1): 1.02[4]
5. Relative vapor density (air = 1): 4.91[5]
6. Critical pressure (MPa): 3.5[6]
7. Octanol/water partition coefficient: 3.87[7]
8. Flash point (℃): 82.22[8]
9. Ignition temperature (℃): 529[9]
10. Explosion limit (%): 5.3[10] sup>
11. Lower explosion limit (%): 0.8[11]
12. Solubility: insoluble in water, soluble in ethanol, ether, etc. Most organic solvents. [12]
13. Refractive index at room temperature (n25): 1.6151
14. Solubility parameter (J· cm-3)0.5: 20.176
15. van der Waals area (cm2·mol– 1): 9.840×109
16. van der Waals volume (cm3·mol-1): 85.110
17. Eccentricity factor: 0.348
18. Liquid phase standard claims heat (enthalpy) (kJ·mol-1): 56.27
19. Liquid phase standard entropy (J·mol-1·K-1): 254.81
20. Liquid phase standard combustion heat (enthalpy) (kJ·mol-1): -5814.04
21. Gas phase standard combustion heat (enthalpy) (kJ·mol-1 ): -5871.37
22. Gas phase standard claims heat (enthalpy) (kJ·mol-1): 113.60
23. Gas phase Standard entropy (J·mol-1·K-1): 377.44
24. Gas phase standard free energy of formation (kJ·mol -1): 214.43
25. Gas phase standard hot melt (J·mol-1·K-1): 159.6
26. Liquid phase standard formation free energy (kJ·mol-1): 193.77
27. Liquid phase standard hot melt (J·mol-1·K-1): 224.4
Toxicological data
1. Acute toxicity[13] LD50: 1840mg/kg (rat oral)
2. Irritation[14] Rabbit transdermal: 0.05ml (24h), moderate irritation.
Ecological data
1. Ecotoxicity[15] LC50: 8.4mg/L (48h) (brown trout, 1 year old, static); 9mg/L (96h) (fathead minnow, static)
2. Biodegradability No data yet
3. Non-biodegradability[16] In the air, when the concentration of hydroxyl radicals is 5.00×105/cm3, the degradation half-life is 7.3h (theoretical).
4. Bioconcentration[17] BCF: 680 (star spot Sichuan plaice, 2 weeks); 205 (red perch, 4h)
Molecular structure data
1. Molar refractive index: 48.92
2. Molar volume (cm3/mol): 139.8
3. Isotonic specific volume (90.2K ): 348.7
4. Surface tension (dyne/cm): 38.7
5. Dielectric constant (F/m): 2.78
6. Polar Chemical rate (10-24cm3): 19.39
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): 3.9
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 0
4. Number of rotatable chemical bonds: 0
5. Number of tautomers:
6. Topological molecular polar surface area (TPSA): 0
7. Number of heavy atoms: 11
8. Surface charge: 0
9. Complexity: 128
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters Number: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. Stability[18] Stable
2. Incompatible substances[19] Strong oxidants, chromic anhydride, chlorate, potassium permanganate
3. Conditions to avoid contact[20] Heating
4. Hazards of aggregation[21] No aggregation
Storage method
Storage Precautions[22] Stored in a cool, ventilated warehouse. Keep away from fire and heat sources. The storage temperature should not exceed 35℃. Keep container tightly sealed. should be kept away from oxidizer, do not store together. Equipped with the appropriate variety and quantity of fire equipment. The storage area should be equipped with emergency release equipment and suitable containment materials.
Synthesis method
1-Methylnaphthalene mainly exists in the naphthalene oil fraction (content 3.45%) and wash oil fraction (content about 5.4%). (1) After the naphthalene oil fraction extracts thiaindene, the residual oil at the bottom of the tower and the washed oil fraction are washed with acid and alkali. After preliminary distillation, the crude methylnaphthalene fraction at 230-270°C is cut out. The cut amount is the raw material. 25-30%, distillation in a distillation tower with no less than 60 theoretical plates, using a reflux ratio of 20:1, and cutting the methylnaphthalene fraction at 240-250°C. Re-distill the methylnaphthalene fraction in a distillation tower with the same tower efficiency, and cut out the crystallizable fraction at 237-241°C and the non-crystallizable fraction at 241-245°C. The crystallizable fraction is frozen to -10~20℃, and 2-methylnaphthalene is prepared by centrifugal separation or press crystallization. Combine the filtrate and the above-mentioned non-crystallizing fractions, wash with 95-98% concentrated sulfuric acid containing 3-5% of the raw material, neutralize with alkali, wash and remove water, and then fractionate on the high-efficiency distillation tower to remove the fractions below 241°C. , remove the crystals by low-temperature freezing, and obtain more than 95% of industrial grade 1-methylnaphthalene. After repeated sulfonation and hydrolysis, a refined product with a purity of 98% can be obtained. (2) High-temperature tar contains about 0.8%-1.2%. After removing the phenol and pyridine alkali from the wash oil fraction at 230-300℃, distill the methylnaphthalene fraction at 240-245℃ and freeze it to -20℃. At this time, β-methylnaphthalene precipitates, and the fraction that does not crystallize at -20°C is the α-methylnaphthalene fraction. After sulfonation and hydrolysis, industrial pure product can be obtained.
Purpose
1. Used as raw materials for organic synthesis of surfactants, water reducing agents, dispersants, drugs, etc. It can be used as a printing and dyeing carrier for polyvinyl chloride fiber and polyester, an emulsifier for BHC, a heat carrier and a solvent, a surfactant, an extractant for sulfur, and can also be used for the production of plasticizers and fiber dyeing auxiliaries. raw material.
2. Standard fuel used to determine the octane number and cetane number of diesel fuel, and also used in organic synthesis.
3. Used in organic synthesis, as printing and dyeing carrier, heat carrier, plasticizer, etc. [23]

 微信扫一扫打赏
微信扫一扫打赏

