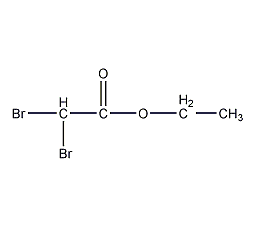
Structural formula
| Business number | 06HX |
|---|---|
| Molecular formula | C4H6Br2O2 |
| Molecular weight | 245.90 |
| label |
None yet |
Numbering system
CAS number:617-33-4
MDL number:MFCD00041718
EINECS number:210-510-7
RTECS number:None
BRN number:None
PubChem ID:None
Physical property data
1. Properties: colorless or light yellow liquid
2. Boiling point (℃, 1.0 mmHg): 194
3. Boiling point (℃, 12 mmHg): 77
4. Density: d 1.9025 g/cm3
5. Solubility: Insoluble in water, slightly soluble in hexane, ether, methylene chloride, acetone, ethyl acetate, ethanol.
6. Relative density (20℃, 4℃): 1.8991
7. Refractive index at room temperature (n20): 1.4633
Toxicological data
None yet
Ecological data
1. Other harmful effects: This substance may be harmful to the environment, and special attention should be paid to water bodies.
Molecular structure data
1. Molar refractive index: 37.77
2. Molar volume (cm3/mol): 124.7
3. Isotonic specific volume (90.2K ): 317.6
4. Surface tension (dyne/cm): 42.0
5. Polarizability (10-24cm3): 14.97
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): 2.2
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 2
4. Number of rotatable chemical bonds: 3
5. Number of tautomers: none
6. Topological molecule polar surface area 26.3
7. Number of heavy atoms: 8
8. Surface charge: 0
9. Complexity: 82.1
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
It has tear-inducing effect. Avoid inhalation and operate in a fume hood.
Storage method
Seal the secret container and store it in a sealed main container in a cool, dry place.
Synthesis method
It is derived from the interaction between ethyl acetate and bromine in the presence of red phosphorus.
Purpose
Under the action of potassium tert-butoxide or highly hindered phenoxide, ethyl dibromoacetate and organoborane undergo an alkylation reaction. α-bromo ester or α,α-dialkyl substituted ester (formula 1)[1]. The use of phenoxy groups (pK 11.7) can reduce self-condensation reactions of starting materials or products. This is an alternative to traditional malonate synthesis methods.
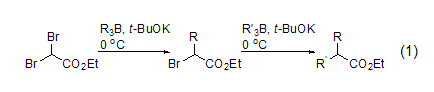
Under low temperature conditions, under the action of zinc/silver-graphite, the Reformatsky reaction of ethyl dibromoacetate occurs to generate α -Bromo-β-hydroxyester (formula 2)[2], then treat α-bromo- with the appropriate group β-Hydroxyesters can give a variety of products. Zinc/silver-carbon reagents can also be used on aldose [3]. Adding diethyl to the Organozinc Reagent in the Reformatsky Reaction Aluminum chloride can improve the reaction to obtainα,β-unsaturated ester, which is an alternative to the Wadsworth-Emmons reaction. Samarium chloride or chromium chloride It can also be used for this reaction (Equation 3)[4].
Underthecatalysisoftriethylborane,theadditionreactionofethyldibromoacetatetoketeneacetalyields1,4-diester(formula4)[5].
Controlling the reaction conditions, benzenethiol can react with ethyl dibromoacetate to generate the corresponding thioacetal (formula 5)[6]. Further alkylation can give pyruvic acid derivatives Object.
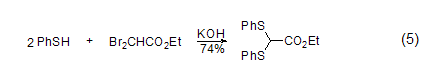
In copper or zinc powder Under the action of ethyl dibromoacetate, monoolefins can undergo ethoxycarbonylcyclopropanation reaction with ethyl dibromoacetate (Formula 6)[7].
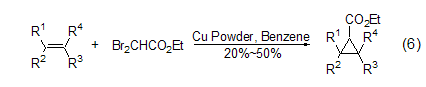

 微信扫一扫打赏
微信扫一扫打赏

