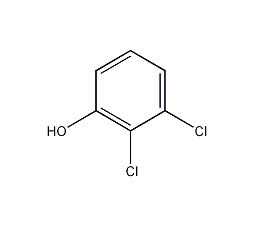
Structural formula
| Business number | 05V5 |
|---|---|
| Molecular formula | C6H4Cl2O |
| Molecular weight | 163 |
| label |
2,3-dichlorophenol, Dichlorophenol, 2,3-Dichlorophenol, 2,3-Dichloro-pheno, Medicine |
Numbering system
CAS number:576-24-9
MDL number:MFCD00002160
EINECS number:209-399-8
RTECS number:SK8450000
BRN number:2043615
PubChem ID:None
Physical property data
1. Physical property data
1. Appearance: white crystal
2. Melting point: 58℃
3. Solubility: soluble in ether and chloroform And hot ethanol, difficult to dissolve in water and benzene
4. Boiling point (ºC): 206
5. Gas phase standard claims heat (enthalpy) (kJ·mol-1 ):-168.0
Toxicological data
None yet
Ecological data
None yet
Molecular structure data
5. Molecular property data:
1. Molar refractive index: 37.92
2. Molar volume (cm3/mol): 111.7
3. Isotonic specific volume (90.2K): 294.0
4. Surface tension (dyne/cm): 47.8
5. Polarizability (10-24cm3): 15.03
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 1
4. Number of rotatable chemical bonds: 0
5. Number of tautomers: 3
6. Topological molecule polar surface area 20.2
7. Number of heavy atoms: 9
8. Surface charge: 0
9. Complexity: 97.1
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. Properties: white crystal.
Storage method
None yet
Synthesis method
2. Preparation method:
Sulfonation of 1,2,3-trichlorobenzene into salt and high-pressure hydrolysis to obtain 3,4-dichloro-
2-Hydroxybenzenesulfonic acid is obtained by hydrolysis with sulfuric acid to remove the sulfonic acid group.
Purpose
3. Usage:
As an organic synthesis intermediate. Used in the synthesis of the drug diuretic acid.

 微信扫一扫打赏
微信扫一扫打赏

